Approximately 40% of biopharmaceutical products rely on sterile fluid management at every stage, from drug development to final fill-finish operations. Inadequate fluid transfer solutions can lead to contamination risks, compromised sterility, and operational inefficiencies.
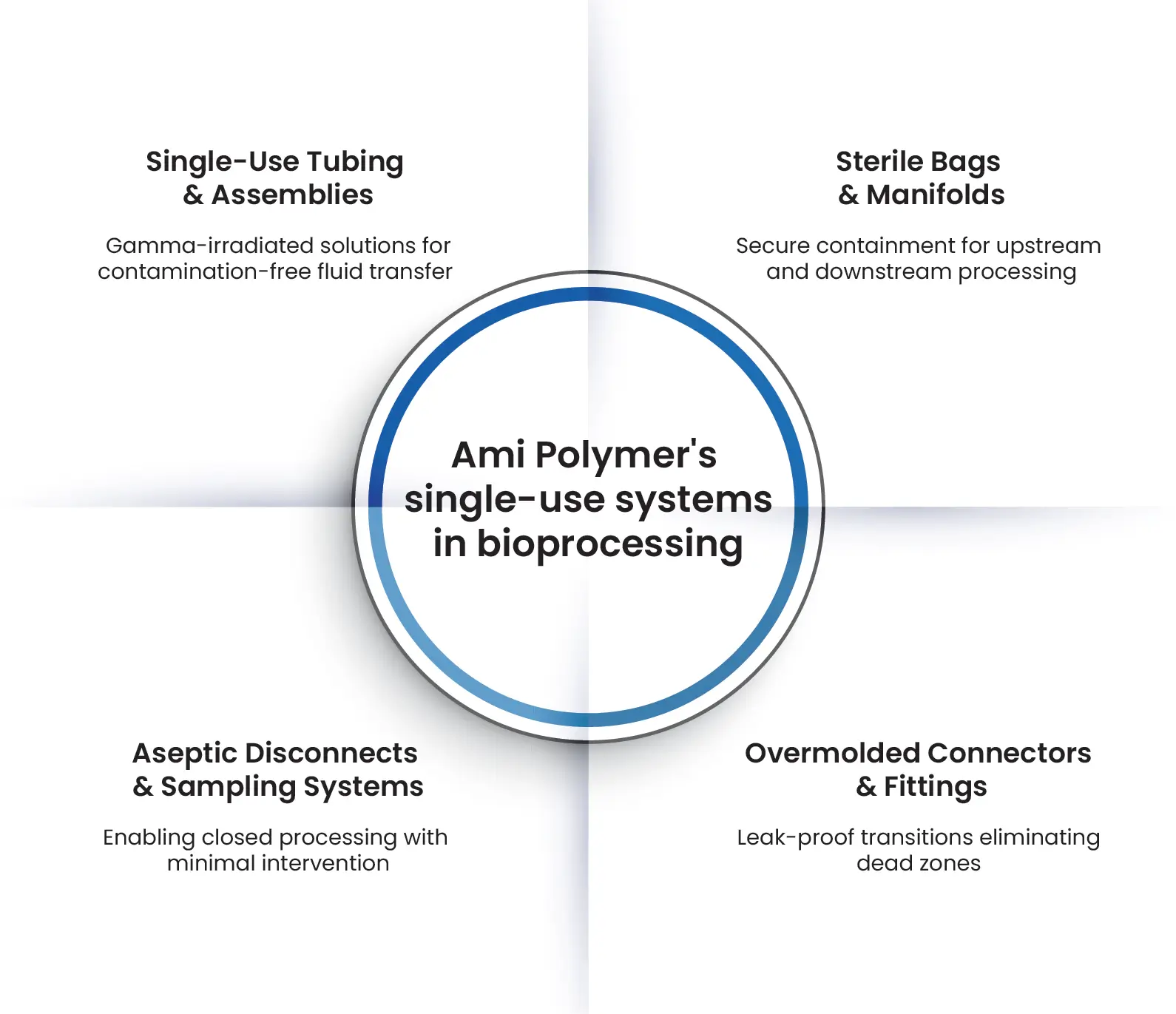
To address these challenges, the industry is shifting towards single-use systems (SUS) for streamlined, contamination-free fluid handling. However, selecting the right components requires expertise in biocompatibility, extractables & leachables (E&L), and regulatory compliance.
At Ami Polymer, we offer a complete portfolio of single-use solutions engineered for sterile fluid transfer, containment, and processing, ensuring seamless integration with biopharma workflows.